
Foundations of theranostics
Radioligand therapy (RLT) and radioligand imaging (RLI) offer a novel, theranostic approach to targeting and treating certain types of cancer.1 This section of the Novartis RLT Institute is designed to help health care professionals (HCPs) understand the basic scientific foundations of RLT. By focusing on the science, we aim to demystify radiation and safety concerns and build confidence in offering patients this approach to precision medicine in cancer.
Radiation science fundamentals
Radiation Overview
Radioactivity is the property of certain unstable atoms (radionuclides) to spontaneously emit radiation. This emission occurs when the nucleus of the atom undergoes radioactive decay, releasing energy or particles.2
Radiation travels through space in 2 distinct forms:
- Photons, which are packets of electromagnetic waves with no mass or electrical charge, travel at the speed of light and are referred to as gamma rays when originating from the nucleus, whereas X-ray photons originate from the orbitals3,4
- Particle radiation, such as alpha particles, beta particles, electrons, or auger electrons, possesses both mass and charge3,5
The activity of radioactive material (RAM) is defined as the number of disintegrations per unit of time.6 A disintegration occurs each time a nucleus emits particles or energy. Activity is measured in a unit called the becquerel (Bq), and 1 Bq equals 1 disintegration per second.6,7
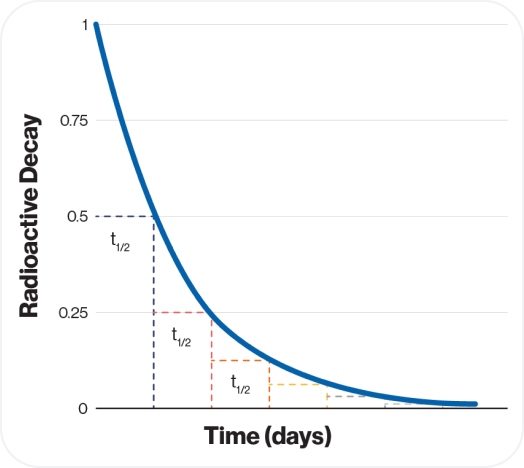
The persistence of this process is characterized by its physical half-life, a measure of how long the radiation continues. Specifically, half-life (𝑡1/2) is the rate of decay of a radionuclide and represents the time necessary for the activity to decay by one-half.8
This graph illustrates the principle of half-life, showing how RAM continually decays over time.
Each radioactive isotope has a unique physical half-life, which directly impacts the shelf life of radioligands and their practical use in medical applications.9
Types of radiation and their role in theranostics
There are 2 types of radiation: non-ionizing and ionizing. Examples of non-ionizing radiation include visible light, ultraviolet light, infrared light, and microwaves.3,10
All radioligands involve ionizing radiation—the forms of ionizing radiation are:
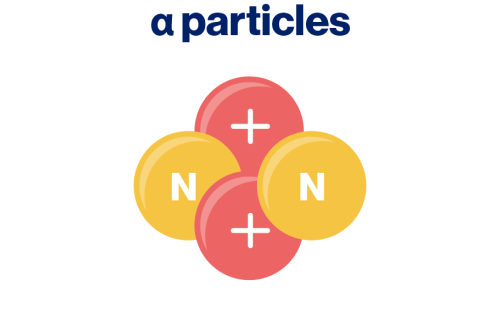
- Emitted by radionuclides during an alpha decay3
- Heaviest, slowest, and highly ionizing, causing high biological damage3
- Low penetration capability (range in tissue from 50-100 µm)3,11
- Used in therapeutic applications12
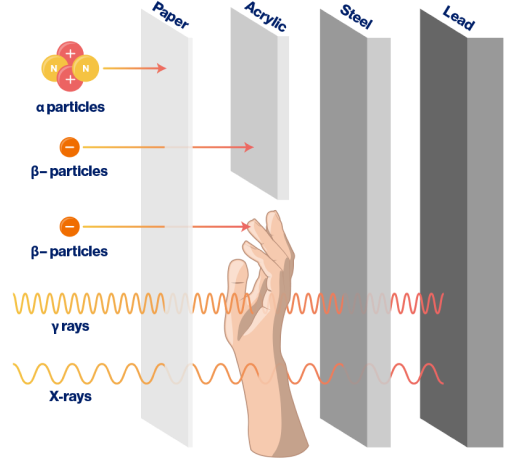
Ionizing radiation can effectively be shielded using different materials, depending on the type of radiation. Heavy particles, such as ɑ particles, can be blocked by a simple sheet of paper. β– particles are best shielded using acrylic, a type of plastic. Low-energy X-rays can often be shielded by steel, while higher-energy X-rays and γ rays require more robust materials such as lead or thick concrete for effective shielding.3,4
How is radiation measured?
To effectively apply ionizing radiation in any medical setting, it is essential to first understand how it is measured. Four key units—radioactivity, exposure, absorbed dose, and dose equivalent—help quantify radiation's intensity and biological effects.
Using the mnemonic R-E-A-D, we can break down these measurements and their practical significance16:
Radioactivity refers to the amount of ionizing radiation released by a material. The units of measure for radioactivity are the curie (Ci) and becquerel (Bq).
For practical purposes, 1 R (exposure) = 1 rad (absorbed dose).16
The dose equivalents differ, depending on the particle type, as some cause more damage than others. For example13:
- Gamma and beta radiation: 1 rad is equal to 1 rem
- Alpha radiation: 1 rad is typically equal to 20 rem


Expert insight
Activity and dose are not the same. Activity refers to the amount of radioactivity in the RLT prescribed, while dose is the actual amount of radiation absorbed in the patient. The administered activity is a factor in determining the dose received by the patient during treatment; other factors include how much is excreted as opposed to bound to the target. For the same administered activity, different patients may have different doses absorbed. Additionally, the same patient may absorb different doses with repeat administrations of the same activity.17
Accurately measuring radiation is critical to ensure the correct amount of radiation is administered to the patient for both diagnostic and therapeutic uses. Additionally, it is important to quantify radiation to inform and monitor exposure levels which enables adherence to safety protocols that protect patients, staff, and the public.18
For more information, visit the Nuclear Regulatory Commission Measuring Radiation website
